Simply stated, yes! Titanium dental implants are safe and effective. They have been used for over 50 years and have been studied extensively over this period. However, that is not to say that there are no risks or complications that jeopardize the success of titanium dental implants. As research and development in dental implant materials continues, there are promising new improvements for dental implant material selection.
Metals are used for dental and orthopedic implants because of their favorable mechanical properties, such as weight-to-strength ratio and good biological performance. However, metallic devices are also prone to corrosion, particularly in the oral environment. Extreme acidic conditions found during inflammation and galvanic corrosion between Titanium implants and other metals in the mouth could greatly affect the mechanical stability and outcome of titanium dental implants.
Implant Corrosion and Reactivity
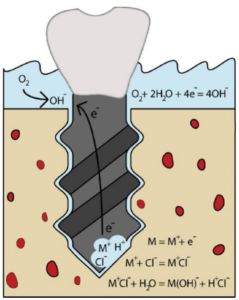
O2 to OH
O2 + 2H2O + 4e = 4OH
M = M+ + e–
M+ + Cl– = M+Cl–
M+Cl- + H2O = M(OH)– + H+Cl–
Dental implants are submerged in ion-rich electrolytes in the body, and have all the requirements for corrosion to take place. Large currents can be induced by the flow of ions and electrons generated during electrochemical reactions occurring between the corroding implant metallic surface and the electrolyte (saliva). Consequently, corrosion events result in the formation of small pits on the surface of the implant that further enhance the corrosive environment around the implant and compromise its stability. Ultimately, this can lead to shortening of the implant’s lifetime and sudden failure. Products of these corrosion reactions can also have cancer causing effects on the tissues surrounding the implant, and contribute to additional causes of rejection or implant failures. Unfortunately, the electrical implications of corrosion on the surrounding tissue have not been extensively investigated.
Passivity of Titanium
Most materials chosen for implant applications exhibit passivity properties and, thus, relatively low corrosion rates compared with those of other more reactive metals, such as zinc, magnesium, or vanadium, which undergo active corrosion even in relatively neutral pH. However, certain environmental conditions can cause corrosion, affecting the mechanical integrity of the implant and the health of the surrounding tissue.
Can Dental Implants react electrically with other metals in the mouth?
Yes, this process is called galvanic corrosion. Galvanic corrosion occurs with direct contact of two dissimilar metals in an electrolytic solution. Galvanic corrosion is not common in dental implant applications because of the presence of only one component, the dental screw, and the insulating nature of the protective passive layer that forms on the surface of implants (oxide layer). Nevertheless, in some individuals, the surrounding tissue could serve as a medium for electrical flow between metallic implants and other types of alloys used in dentistry for amalgams or orthodontic devices.
The electrical implications of corrosion and its effect on the surrounding tissue may be an important key to this puzzle, but such effects still remain unclear. Corrosion events generate electrical currents and consequently, it is fair to suggest that cells and tissues in individuals with implants are exposed to abnormal electrical signals for extended periods of time. Bone cells are sensitive to electrical signals and, thus, could be strongly affected by these corrosion currents. Moreover, these abnormal electrical signals may provide an alternate explanation for the unresolved causes of inflammatory complications and eventual implant loosening.
Conclusion
In summary, here are the main points that you should take from this article:
- Titanium dental implants have great long-term success and low chance of complications.
- However, there still are risks including corrosion that could affect the implant and/or even cause it to fail.
- Although titanium implants could potentially react electrically with other metals in the mouth, the long-terms effects remain unclear.
As always, let us know your questions in the comments below!

Leave a Reply